Equations and Formulas in Chemistry
|
Since every compound is made up different combinations of atoms, there is a way to represent every compound's atomic structure using letters and numbers. These are called Chemical Formulas. They are formed using the abbreviations for the elements that make it up plus the amounts of those elements as represented by subscripts or coefficients.
An example of a Chemical Formula is H2O. In case you didn't know, this is the chemical formula or water. It means that there are two atoms of hydrogen and one atom of oxygen for every molecule of water. The subscript (lower and smaller) two after the H indicates that there are two hydrogen molecules, as hydrogen is abbreviated H. Also, since there is no subscript after the O, this indicates that there is only one oxygen atom in one molecule.
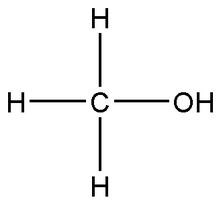
There are many different ways of representing chemical formulas. These include empirical formulas, molecular formulas, and structural formulas. Each of these is a different way of expressing the same formula, although it is shown differently. Structural Formulas show the structure of the bonding between the atoms in a compound visually. There are many different types of structural formulas, including the one shown at right and three dimensional ones as well. Molecular Formulas show the number of each kind of atom in each molecule of a substance. An important difference between them and empirical formulas is that if a molecule has two carbon molecules and four hydrogen molecules the equation of C2H4 must be used. It can't be condensed down to CH2 by dividing all the subscripts by two like it can be in an empirical formula because the amount of atoms in each molecule can be higher than the lowest whole-number ratio. Ionic Equations demonstrate reactions between aqueous ions in a solution. All reactants are converted into charged ions which are in turn treated as individual reactants. For more information on ionic and net ionic equations, see the Ionic Bonds page. Empirical Formulas are mostly the same as molecular formulas, except that they only show the ratios of the numbers of each of the kinds of elements present in a compund. For example, the molecular formula C9H18 is written as CH2 as an empirical formula because when both subscripts are divided by 9 they can be reduced to this small ratio of whole numbers. Empirical formulas are mostly used for ionic compounds where there are no molecules, only formula units and the ratios of each kind of element present in the compound can be represented as small whole numbers. |
NaCl, CaF2 and UF6 are all examples of empirical formulas for ionic compounds. They indicate the ratios of the different kinds of atoms present in the ionic compound formed from each formula. They each represent one formula unit for the ionic compound. See Ionic Bonding for more information.
CO2, COH4, and O3 are all examples of molecular formulas. They represent the amount of atoms of each element that are present in each molecule of the substance produced. So for CO2 (Carbon Dioxide) there are two atoms of oxygen and one atom of carbon in every molecule of carbon dioxide.
|