Grams to Moles and Moles to Grams Conversion
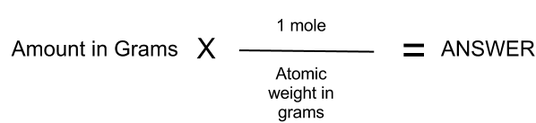
Here's some basic info to start you off on this topic: A mole is a unit representing the amount of particles of Carbon-12 in 12 grams of Carbon-12. This number is represented by Avagadro's Number, which estimates to 6.02X10^23. The mole to gram ratios of different substances depend on their atomic weights and their weight in grams. To calculate the amount of grams of a substance given its atomic weight and number of moles, set up an equation like this:
So, to convert from grams to moles, divide the amount in grams by the atomic weight in grams to find the amount of moles. The grams cancel, because they are both on the bottom of the equation if you make the amount in grams a fraction over 1 gram which leaves moles and the answer. So, for example, 5 grams of Potassium (Atomic weight 39) equals .128 moles of Potassium. To find the number of particles in .128 moles of potassium, simply multiply .128 by Avacado- I mean Avagadro's constant. This yeilds 77056000000000000000000, or 77 sextillion particles. A lot of atoms, and only in 5 grams.
More coming soon!