Covalent Bonding
Covalent Bonding is the most common and, in my opinion, most important kind of bonding. It is a kind of bonding between atoms within a molecule and forms some of the strongest bonds anywhere.
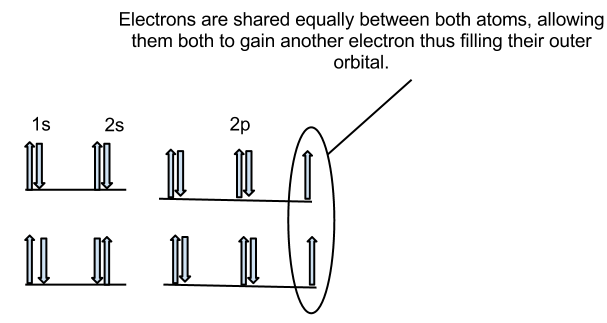
The principle of a covalent bond is that atoms want to fill their Valence Shell, or outermost energy level with the largest amount (8) of electrons. For example, Fluorine (F on the periodic table) has 7 electrons in its valence shell. Chlorine wants one electron to fill its valence shell and so does Fluorine. In Orbital notation, the electrons will look like this:
The principle of a covalent bond is that atoms want to fill their Valence Shell, or outermost energy level with the largest amount (8) of electrons. For example, Fluorine (F on the periodic table) has 7 electrons in its valence shell. Chlorine wants one electron to fill its valence shell and so does Fluorine. In Orbital notation, the electrons will look like this:
As you can see, the last electrons in the p suborbitals of both atoms are shared with eachother, thus filling their valence shells. This is how it looks in Electron-Dot Notation:
The human body is made up almost entirely of covalent bonds, as it is 70% water (a covalent compound) and also a lot of organic carbon compounds (mostly covalent).
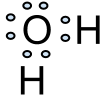
For atoms that make multiple bonds such as oxygen which makes two bonds, for example, two or more electrons can be shared between one or more other atoms. Hydrogen atoms are one of the few exceptions to the octet rule, which states that all atoms want 8 electrons in their valence shell to reach their most stable state. So in water, which is two hydrogens bonded to one oxygen, the two bonding sites of water are both taken up by a hydrogen. Take a look at the electron dot diagram below:
For atoms that make multiple bonds such as oxygen which makes two bonds, for example, two or more electrons can be shared between one or more other atoms. Hydrogen atoms are one of the few exceptions to the octet rule, which states that all atoms want 8 electrons in their valence shell to reach their most stable state. So in water, which is two hydrogens bonded to one oxygen, the two bonding sites of water are both taken up by a hydrogen. Take a look at the electron dot diagram below:
Using this method, large molecules can be build up by, for example, chaining up carbon atoms connected to hydrogens on the side to form many of the plastics, fats, oils, and other substances we see in daily life. (They're called hydrocarbons, by the way).
There are two types of covalent bonds: Polar Covalent Bonds and Nonpolar Covalent Bonds. The type of bond is determined by the electronegativity of the two atoms that are being bonded. Every element has a certain electronegativity, which is a measure of how much atoms attract electrons in a bond. The electronegativities generally increase from left to right as atoms gain more electrons in the same energy level. This is partly because they gain more protons which creates a stronger attraction between them and the electron cloud of the bonded atom. Anyway, if the electronegativity difference between the two atoms is less than .5, then the bond is considered nonpolar as the electrons are distributed fairly evenly between the two atoms in the bond. If the electronegativity is between .5 and 1.7, then the bond is considered polar covalent as one atom pulls obviously stronger on the electrons than the other. For electronegativity differences of higher than 1.7, the bond is considered ionic.
Sometimes atoms make multiple bonds with eachother. They do this if they have two missing electrons and another atom can share two electrons with them in a covalent bond. For example, oxygen molecules consist of two oxygen atoms covalently bonded together with a double bond. This way they share two of their electrons which provides them with enough electrons to fill both their octets. See the diagram below for an example of a double bond:
As you can see, the atoms turn so that their two open sides face eachother, creating a double covalent bond between them as both open spots in their energy levels fill eachother.
Resonance is a phenomenon that is in involved with molecules that have bonds that constantly alternate, or resonate, from being double bonds to single bonds. An example is Ozone (03), in which there is one double bond and one single bond. However, the bonds are technically neither double or single, rather a combination of both. You could call it a 1.5 bond. Instead of going around just two of the oxygen atoms, the electrons move around all three of the atoms. It's a bit hard to understand, but the most important thing to remember is that they can't be represented by just one diagram, but rather both alternate forms have to be shown like below:
Notice that in the top of the above diagram, the rightmost oxygen ion shares a double bond with the middle oxygen, while in the bottom one the left oxygen atom shares the double bond. In all reality, neither share the bond at any given moment and the positions of the electrons varies constantly at the speed of light. Just remember that it's not on either side, but in between.